
In an interview during the annual Steering Committee meeting, Drs. Rosa Gonzalez-Guarda and Cherise Harrington, cochairs of the Health Equity Core Working Group, shared insights and next steps for health equity in the NIH Pragmatic Trials Collaboratory.
One question discussed at the meeting was how to embed health equity in pragmatic clinical trials. Gonzalez-Guarda said that a key component of addressing health equity is acknowledging the structural and social drivers of that inequity and developing strategies to target them.
“The next phase is to figure out ways that we can encourage embedded pragmatic clinical trials that address the structural causes of health inequities,” said Gonzalez-Guarda, who noted that the National Institutes of Health is supporting more structural interventions.
One way the NIH Pragmatic Trials Collaboratory can lead is by integrating a pragmatic clinical trial lens into structural intervention work, Gonzalez-Guarda said. This work is central to the Health Equity Core, which was launched last fall and began meeting regularly in early 2023.
Health Equity Core members are reviewing a checklist on how to infuse a health equity lens into the research life cycle. As a next step, Harrington and Gonzalez-Guarda would like to ask Core members to think critically about how the checklist can be adapted for pragmatic clinical trials and develop it as a new tool for the research community.
Harrington said another way the Health Equity Core can make an impact is by gathering examples from the NIH Collaboratory Trials of data on underrepresented populations that are not statistically significant but may be clinically significant so that other projects can learn from them.
Drawing from and adding to the NIH Pragmatic Trials Collaboratory’s lessons learned is also a key strategy for the Health Equity Core’s work. Harrington said it is important to share knowledge outside of the research community.
“We need to remember to ask ourselves, 'What am I leaving with this community after the funding is gone that they can still leverage?,’” said Harrington.
Many of the NIH Collaboratory Trials are already implementing health equity plans and engagement strategies to reach diverse patient populations, providers, and community stakeholders.
Gonzalez-Guarda said that it is important to continue this work and to support the careers of junior researchers working on pragmatic clinical trials so they can use engagement practices and be successful. In addition to preparing investigators to engage with diverse communities, part of the engagement work may also be training for communities to better engage with projects in a more meaningful way, Harrington said.
Developing tools and resources is a top priority as the Health Equity Core moves forward, and Gonzalez-Guarda and Harrington look forward to providing guidance for pragmatic clinical trials and continuing the conversation from the Steering Committee meeting.
Learn more about the Health Equity Core and see the complete materials from the 2023 Steering Committee meeting.







 In an interview at the NIH Pragmatic Trials Collaboratory’s annual Steering Committee meeting and 10th anniversary celebration, we asked Dr. Liz Turner and Dr. Patrick Heagerty to reflect on the role of the
In an interview at the NIH Pragmatic Trials Collaboratory’s annual Steering Committee meeting and 10th anniversary celebration, we asked Dr. Liz Turner and Dr. Patrick Heagerty to reflect on the role of the 
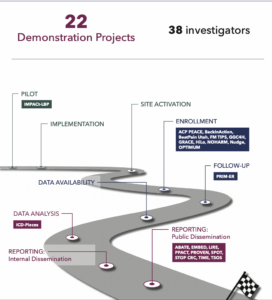
 IMPACt-LBP, currently in the 1-year planning phase, will evaluate implementation of the American College of Physicians guideline for low back pain, which involves multidisciplinary collaborative care that includes doctors of chiropractic and physical therapists. The study will measure the effects of first-contact patient referral to these clinicians on physical function, pain, opioid prescriptions, and other patient-level outcomes. The study is administered by the National Center for Complementary and Integrative Health with additional support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the National Institute of Child Health and Human Development. The project is led by Drs. Christine Goertz, Adam Goode, and Hrishikesh Chakraborty of Duke University and Dr. Jon Lurie of Dartmouth Hitchcock Medical Center.
IMPACt-LBP, currently in the 1-year planning phase, will evaluate implementation of the American College of Physicians guideline for low back pain, which involves multidisciplinary collaborative care that includes doctors of chiropractic and physical therapists. The study will measure the effects of first-contact patient referral to these clinicians on physical function, pain, opioid prescriptions, and other patient-level outcomes. The study is administered by the National Center for Complementary and Integrative Health with additional support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the National Institute of Child Health and Human Development. The project is led by Drs. Christine Goertz, Adam Goode, and Hrishikesh Chakraborty of Duke University and Dr. Jon Lurie of Dartmouth Hitchcock Medical Center.  ACP PEACE, currently in the implementation phase, is testing an advance care planning program that combines clinician communication skills training and patient video decision aids. ACP PEACE is focused on patients with advanced cancer and their clinicians in oncology settings. The study is administered by the National Institute on Aging and is led by Drs. Angelo Volandes and James Tulsky of Harvard Medical School.
ACP PEACE, currently in the implementation phase, is testing an advance care planning program that combines clinician communication skills training and patient video decision aids. ACP PEACE is focused on patients with advanced cancer and their clinicians in oncology settings. The study is administered by the National Institute on Aging and is led by Drs. Angelo Volandes and James Tulsky of Harvard Medical School.  SPOT, a recently completed pragmatic trial that published its main outcomes earlier this year, compared 2 low-intensity outreach programs—care management and dialectical behavior therapy skills training—to usual care in adults at risk of self-harm or suicidal behavior. The study was administered by National Institute of Mental Health and was led by Dr. Greg Simon of the Kaiser Permanente Washington Health Research Institute.
SPOT, a recently completed pragmatic trial that published its main outcomes earlier this year, compared 2 low-intensity outreach programs—care management and dialectical behavior therapy skills training—to usual care in adults at risk of self-harm or suicidal behavior. The study was administered by National Institute of Mental Health and was led by Dr. Greg Simon of the Kaiser Permanente Washington Health Research Institute. 
 Much of this content is housed in Rethinking Clinical Trials
Much of this content is housed in Rethinking Clinical Trials Anne Trontell
Anne Trontell
 Partha Bhattacharyya, Program Director for the National Institute on Aging and a lead developer of the National Institute on Aging IMbedded Pragmatic Alzheimer’s disease and AD-Related Dementias Clinical Trials (
Partha Bhattacharyya, Program Director for the National Institute on Aging and a lead developer of the National Institute on Aging IMbedded Pragmatic Alzheimer’s disease and AD-Related Dementias Clinical Trials ( Wendy Weber
Wendy Weber Gregory Simon
Gregory Simon On what has surprised him the most – “Patients and clinicians pretty readily grasp the need and the concept. They want to do it. The data part is a lot harder than most people thought. The technology has come a long way; we can do it now. Probably the hardest thing has actually been that the business models for health systems run counter to generating evidence. It’s something that we really haven’t overcome at this point.”
On what has surprised him the most – “Patients and clinicians pretty readily grasp the need and the concept. They want to do it. The data part is a lot harder than most people thought. The technology has come a long way; we can do it now. Probably the hardest thing has actually been that the business models for health systems run counter to generating evidence. It’s something that we really haven’t overcome at this point.”