In an interview at the Steering Committee’s annual meeting in April, Dr. Wendy Weber, Dr. Josephine Briggs, and Dr. Catherine Meyers celebrate the success of the NIH Pragmatic Trials Collaboratory and look at the roadmap to its future.
In the Beginning…
The NIH Pragmatic Trials Collaboratory (then the NIH Health Care Systems Research Collaboratory) began as an NIH Common Fund–initiated program. According to Dr. Josephine Briggs, Director Emeritus of the National Center for Complementary and Integrative Health (NCCIH), the idea began with a recognition that “explanatory trials were often run in ways to demonstrate that an intervention worked with selected populations implemented in a very controlled environment… We really needed to test those interventional trials and see whether they really worked when generalized to a broader population likely to be using them to settings in which the intervention implementation wasn’t as tightly controlled.”
In 2012, the Common Fund awarded both the Coordinating Center and the first 7 NIH Collaboratory Trials (ABATE, BPMedTime, LIRE, PPACT, SPOT, STOP CRC, TiME).
The Present
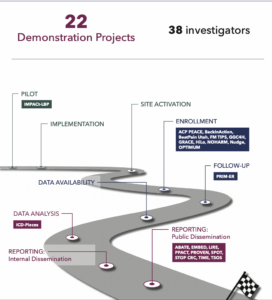
In its first 10 years, the NIH Pragmatic Trials Collaboratory has supported 22 NIH Collaboratory Trials involving 38 investigators.
Dr. Cathy Meyers, Director Emeritus of the NCCIH Office of Clinical and Regulatory Affairs stated that “The Coordinating Center has really been a scribe throughout this last decade of recording problems and, more importantly, their solutions, and has even, with the last wave of solicitations for applications, incorporated training.”
 Much of this content is housed in Rethinking Clinical Trials®: A Living Textbook of Pragmatic Clinical Trials, a free online textbook developed and maintained by the NIH Collaboratory. Drawing on experiences from the program’s NIH Collaboratory Trials and community of experts, the Living Textbook provides guidance on each step of a pragmatic trial, from study design through applying results in the real world. The Living Textbook is continually updated and expanded as new information emerges. This new knowledge is also disseminated through the NIH Collaboratory’s Knowledge Repository and weekly Grand Rounds series.
Much of this content is housed in Rethinking Clinical Trials®: A Living Textbook of Pragmatic Clinical Trials, a free online textbook developed and maintained by the NIH Collaboratory. Drawing on experiences from the program’s NIH Collaboratory Trials and community of experts, the Living Textbook provides guidance on each step of a pragmatic trial, from study design through applying results in the real world. The Living Textbook is continually updated and expanded as new information emerges. This new knowledge is also disseminated through the NIH Collaboratory’s Knowledge Repository and weekly Grand Rounds series.
Looking to the future
“There are now 8 different partnering institutions and centers that support the NIH Pragmatic Trials Collaboratory, and we are open to more,” said Dr. Wendy Weber, NCCIH Project Officer for the NIH Collaboratory.
When asked what she is looking forward to in the future, Dr. Weber stated that “There is a unique opportunity to look at trials that address health disparities. How do we solve what we know is a problem in the healthcare system? How can we design interventions to improve that. We do so much research documenting how much disparity there is. Let’s do something to actually improve that.”
Towards this end, the NIH released a request for applications (RFA) for new NIH Collaboratory Trial grants that address health disparities. The RFA for this funding opportunity encourages applications that focus on improving health outcomes in populations that experience health disparities, such as higher rates of disease or mortality compared with the general population. Applications are due June 17, 2022. Letters of intent are due 30 days prior (May 17, 2022).
We will be sharing more insights form the 2022 NIH Pragmatic Trials Steering Committee Meeting over the coming weeks. All meeting materials are available online.
View the full interview.
About the NIH Pragmatic Trials Collaboratory
The NIH Pragmatic Trials Collaboratory Coordinating Center consists of the Duke Clinical Research Institute, the Harvard Pilgrim Health Care Institute, the Kaiser Permanente Washington Health Research Institute, the Johns Hopkins Berman Institute of Bioethics, the University of Washington, and the University of Michigan. The program is supported by the National Institutes of Health (NIH) through cooperative agreement U24AT009676 from the Office of Strategic Coordination within the Office of the NIH Director. It is also supported by the NIH through the NIH HEAL Initiative under award number U24AT010961. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or its HEAL Initiative. To learn more about the program, visit rethinkingclinicaltrials.org.





 Investigators from the NIH Pragmatic Trials Collaboratory in 2023 shared study results, generated new knowledge, and developed innovative methods in the design, conduct, and analysis of pragmatic clinical trials. Their work included insights from the Coordinating Center and
Investigators from the NIH Pragmatic Trials Collaboratory in 2023 shared study results, generated new knowledge, and developed innovative methods in the design, conduct, and analysis of pragmatic clinical trials. Their work included insights from the Coordinating Center and  Investigators from the NIH Pragmatic Trials Collaboratory in 2022 shared study results, generated new knowledge, and developed innovative research methods in pragmatic clinical trials. Their work included insights from the Coordinating Center and
Investigators from the NIH Pragmatic Trials Collaboratory in 2022 shared study results, generated new knowledge, and developed innovative research methods in pragmatic clinical trials. Their work included insights from the Coordinating Center and 
 Much of this content is housed in Rethinking Clinical Trials
Much of this content is housed in Rethinking Clinical Trials