
In a new episode of the NIH Collaboratory Podcast, Drs. Christopher Lindsell and Adrian Hernandez discussed the need for virtual vigilance in decentralized trials.
Listen to the podcast. For alerts about new episodes, subscribe for free on Spotify, Amazon Music, Apple Podcasts, or SoundCloud.
Decentralized trials move research activities from a centralized location, such as a hospital, to a location that is closer to the patient, such as their home or their local pharmacy. In addition to being more convenient for participants, decentralized trials provide research teams with the opportunity to reach a broader population.
Lindsell and Hernandez co-authored a JAMA Viewpoint entitled “Ensuring Virtual Vigilance in Decentralized Clinical Trials.” They also collaborated on the recently published Living Textbook Chapter on Decentralized Pragmatic Clinical Trials. In the podcast, they explored the opportunities that decentralized trials present and the reasons why monitoring is critical to obtaining high-quality results.
“There are 4 areas we have to think of really deeply,” Lindsell said, “Do we have the right person enrolled in the study and giving us the data? Did we get them the right intervention? And are they adherent and are they following the treatment in the right way? Are they giving us the right data and are we reacting to those data in a timely and an appropriate way?”
Research teams conducting decentralized trials should take advantage of different data streams, including metadata, to characterize ongoing engagement and ensure the appropriate engagement of participants throughout a study.
Lindsell is professor and cochief of biostatistics and bioinformatics, director of data science and biostatistics at the Duke Clinical Research Institute, and director of biostatistics and bioinformatics at the Duke Clinical and Translational Science Institute—all at Duke University. Hernandez, who serves as co–principal investigator of the NIH Pragmatic Trials Collaboratory Coordinating Center, is a professor of medicine and vice dean in the Duke University School of Medicine and executive director of the Duke Clinical Research Institute.





 In this Friday’s Rethinking Clinical Trials Grand Rounds, Jeffrey Spaeder and Adrian Hernandez will present
In this Friday’s Rethinking Clinical Trials Grand Rounds, Jeffrey Spaeder and Adrian Hernandez will present 
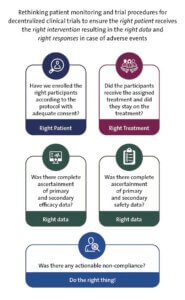 “With these risks to integrity forefront, and consistent with the principles of using decentralized elements and risk-proportionate monitoring, we propose [decentralized trials] frame their monitoring and oversight to ensure the right patient receives the right intervention, contributes the right data, and that the right response occurs for adverse events or noncompliance,” the authors wrote.
“With these risks to integrity forefront, and consistent with the principles of using decentralized elements and risk-proportionate monitoring, we propose [decentralized trials] frame their monitoring and oversight to ensure the right patient receives the right intervention, contributes the right data, and that the right response occurs for adverse events or noncompliance,” the authors wrote.
 In this Friday’s PCT Grand Rounds, Adrian Hernandez and Christopher Lindsell of Duke University will present
In this Friday’s PCT Grand Rounds, Adrian Hernandez and Christopher Lindsell of Duke University will present