Speaker
Michael D. Kappelman, MD, MPH
Professor, Pediatric Gastroenterology
University of North Carolina at Chapel Hill
Keywords
PCORnet; PCORI; Inflammatory Bowel Disease; Pediatrics; Common Data Model; Study Design
Key Points
- Inflammatory Bowel Disease (IBD) is a chronic gastrointestinal condition affecting roughly 100,000 youth in the United States. It has a profound impact on nutrition, growth, physical, and psychosocial development. Anti-TNF biologics are the only FDA-approved advanced therapies for children, and approximately 30% of patients experience treatment failure within 2 years. There’s an urgent need for comparative effectiveness research that can guide treatment decisions when anti-TNF fails.
- COMPARE-Pediatrics IBD, a PCORnet® study, includes 2 parallel multi-center, prospective cohort studies and retrospective cohort studies. The former, developed with multi-stakeholder input, will compare the effectiveness of emerging therapies in children with IBD; the latter will characterize the safety of these treatments and explore the heterogeneity of treatment effects across subgroups.
- The study is utilizing PCORnet’s® infrastructure, including Prep-to-Research Queries and the PCORnet® Common Data Model (CDM), to inform the study design; identify administrative efficiencies; support recruitment; ease site burden; assess representativeness of the study population; and otherwise bolster their research.
Discussion Themes
Planning a PCORnet® study is a lot of work (and takes time). Start the process early and know that benefits may be on the back-end.
The study team opted not to conduct a randomized pragmatic trial because they anticipated that desperate families would be reluctant “roll the dice” with randomization and because insurance coverage for expensive off-label medications often dictates which therapy a patient can receive.
While the CDM is effective for structured data (like labs and diagnoses), Dr. Kappelman noted it cannot yet capture nuanced interpretations, such as specific MRI findings, which require more advanced AI or manual review.




 The NIH and the Patient-Centered Outcomes Research Institute (PCORI) are major funders of pragmatic clinical trials embedded in healthcare systems. An existing chapter of the Living Textbook of Pragmatic Clinical Trials provided guidance on how to
The NIH and the Patient-Centered Outcomes Research Institute (PCORI) are major funders of pragmatic clinical trials embedded in healthcare systems. An existing chapter of the Living Textbook of Pragmatic Clinical Trials provided guidance on how to 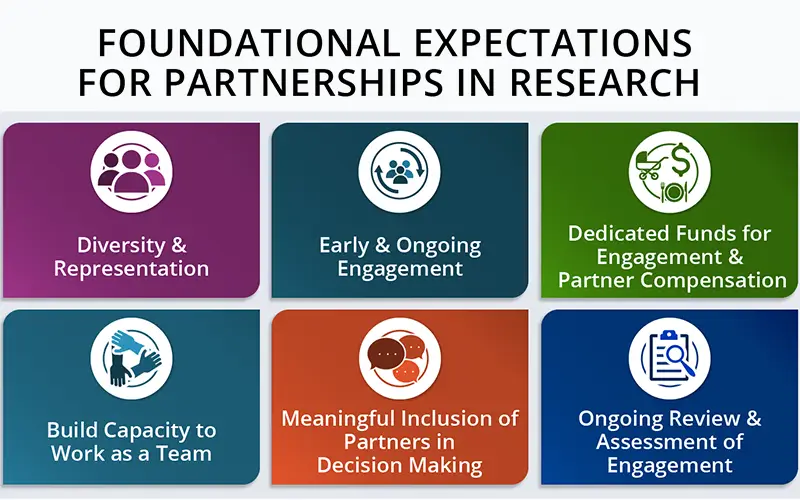 The Patient-Centered Outcomes Research Institute (PCORI) announced the release of its new Foundational Expectations for Partnerships in Research. The resource is an update of the 2014 PCORI Engagement Rubric. It builds on a growing body of evidence about engaging patients and other partners in patient-centered comparative effectiveness research.
The Patient-Centered Outcomes Research Institute (PCORI) announced the release of its new Foundational Expectations for Partnerships in Research. The resource is an update of the 2014 PCORI Engagement Rubric. It builds on a growing body of evidence about engaging patients and other partners in patient-centered comparative effectiveness research.

 In this Friday’s PCT Grand Rounds, Erin Holve, Russell Rothman, Schuyler Jones, and Neha Pagidipati will present
In this Friday’s PCT Grand Rounds, Erin Holve, Russell Rothman, Schuyler Jones, and Neha Pagidipati will present