The NIH Pragmatic Trials Collaboratory held its 2026 Annual Steering Committee Meeting in Bethesda, Maryland, on April 13 and 14.
Program leaders discussed new methods and areas of focus for pragmatic trials, the value of incorporating qualitative research methods into these studies, and the importance of in-depth feasibility assessment in the planning phase. Presenters shared lessons from their new, ongoing, and completed NIH Collaboratory Trials and shared strategies for boosting the efficiency, relevance, and impact of their work.
All materials from the meeting are now available online.
The meeting began with a session moderated by former FDA Commissioner Robert Califf to consider what is on the horizon for the NIH Pragmatic Trials Collaboratory. Panelists discussed the priority evidence that can be addressed through pragmatic trials embedded in healthcare systems and the methods and knowledge required for the next generation of these studies.
Califf joined NIH Collaboratory Coordinating Center co–principal investigator Kevin Weinfurt after the session for an interview.
“When we started the [NIH Collaboratory], you could see that the technology was going to develop so that we could use electronic health records, claims data, and patient-derived data to answer a lot more questions more quickly,” Califf recalled. Califf led the research team that responded to the request for applications that prompted the formation of the NIH Pragmatic Trials Collaboratory program in 2012.
“I don’t think technology is any longer the limitation,” Califf said, “it’s really how we work together as people to make this machine work quickly to answer questions that are critical to the health of the population.”
“We’ve got a really fragmented, dynamic healthcare system,” added Weinfurt, “and alignment with leadership in those health systems to identify important questions is something we’re going to have to keep tracking and learning how to do better as that environment changes.”
The NIH Pragmatic Trials Collaboratory added 2 new trials in the past year. CARNATION will test a multicomponent implementation support intervention to enable community health centers’ systematic use of electronic health record technologies for coordinating primary care–based pain care. The EquiP PC trial will test an adapted behavioral health integration toolkit to improve chronic pain care. Both projects are supported through the NIH HEAL Initiative by grants from the National Institute of Neurological Disorders and Stroke.
Learn more about the NIH Collaboratory Trials.
In the coming weeks, we will share more highlights from the 2026 Annual Steering Committee Meeting. Access the complete meeting materials.






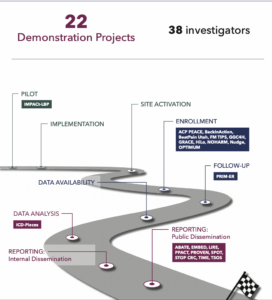
 Much of this content is housed in Rethinking Clinical Trials
Much of this content is housed in Rethinking Clinical Trials In this Friday’s PCT Grand Rounds, Dr. Jeremy Sugarman, Dr. Stephanie Morain, and Juli Bollinger of Johns Hopkins University and Dr. Kevin Weinfurt of Duke University will present
In this Friday’s PCT Grand Rounds, Dr. Jeremy Sugarman, Dr. Stephanie Morain, and Juli Bollinger of Johns Hopkins University and Dr. Kevin Weinfurt of Duke University will present