Developing a Compelling Grant Application
Section 2
Find the Right Program Official and Study Section
The NIH is a major funder of health research and comprises 27 institutes and centers (ICs), each with its own mission, budget, scientific priority, funding strategy, and staff (including those who work with applicants, such as program officials, scientific review officers, and grants management officers).
Early in the planning of developing any application, including those for a pragmatic trial, NIH recommends contacting a program official at one of the ICs whose mission most closely matches the trial. The purpose of this contact is to talk to a program official who can gauge an IC's potential enthusiasm for a research idea, recommend which notice of funding opportunity (NOFO) to use for the application, and provide guidance to the investigator both pre-submission and post-review. Because finding the right program official is a critical first step in developing a successful grant application, the NIH Research Portfolio Online Reporting Tools (RePORTER) website has a matchmaker function. Through the site, prospective grant applicants can enter up to 15,000 words, such as the proposed application’s project aims, abstracts, or other scientific text, and search for a "similar project" and a "similar program official."
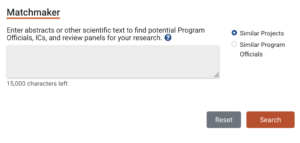
Searching for a program official produces a list of people and their affiliated IC, email addresses, and number of projects. For example, the search “pragmatic clinical trial” produced a list of 191 program officials.
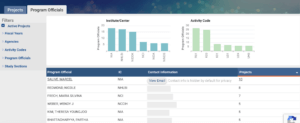
After researching the program officials and ICs, prospective investigators can select one with projects most closely matching the proposed application and then email the program official with
- A 1- to 2-page draft of specific aims for the proposed application
- A request to discuss the proposed application and whether it is a fit with the IC's mission and priorities
- A request to provide the name of another colleague or IC if the proposed application is not a fit with their portfolio.
The program official will be in touch to review the specific aims. If the program official is not a right fit for a project, they may recommend someone who might be more suitable or recommend that the investigator contact a program official at another IC. In some cases, more than one program official is potentially appropriate, such as when a project would fit within a disease-specific and non–disease-specific IC. If so, investigators may contact both program officials.
Applications at NIH are reviewed by groups of scientists from the research community that are called study sections. The NOFO will indicate how the application will be reviewed. If the NOFO states that the application will be evaluated for scientific and technical merit by an appropriate Scientific Review Group convened by a specific IC, then the application will be reviewed by that specific IC. Many NOFOs do not specify an IC to conduct the review, and in those cases, the application will be assigned to a standing study section in the Center for Scientific Review (CSR). Applicants can request their application be assigned to a specific study section at the CSR. To help investigators find the study section with the most appropriate scientific expertise for reviewing their grant, the NIH has developed the Assisted Referral Tool.
The Assisted Referral Tool (ART) was developed by the CSR to recommend potentially appropriate study sections, based on the scientific content of a user’s grant application. The information you provide to the tool is only used to recommend study sections and is not stored. ART results should be discussed with the program official, if possible, to help narrow down which study section to request when submitting the application.
How to Use the ART
- Select your mode of operation based on your search goals.
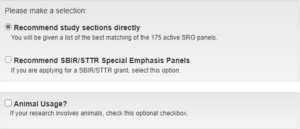
- Enter application text and/or title into the appropriate text boxes. Concepts from the Research, Condition, and Disease Categorization Thesaurus are extracted and weighted against relative frequency of concepts within the text. Concepts found within the title receive an additional influence on the search results.

- The search results are ranked based upon relevancy, and the number of results within each relevancy category is determined by multiple criteria. Within each relevancy category, the results are ordered alphabetically based on Scientific Review Group abbreviation.

Examples of Using the ART
- Using the NIH Collaboratory Trial BackInAction as an example, we first select “Recommend study sections directly.” Next, input the title and summary in the appropriate text boxes, as shown below.

- Search results will appear. In this example, the top 3 strongest results are ASG, BMHO, and MRS. These represent the most relevant sections based on concepts found in the search input.

SECTIONS
DISCLAIMER
The views expressed in this chapter should not be interpreted as representing the official views of the US Department of Health and Human Services, the National Institutes of Health, the National Institute on Aging, the National Center for Complementary and Integrative Health or PCORI, except where noted.
ACKNOWLEDGEMENT
The authors thank Dr. Tracy Wang, the chief officer for Comparative Clinical Effectiveness Research (CER) at the Patient-Centered Outcomes Research Institute (PCORI), for her review of the content related to PCORI.



