Patients who participated in a cognitive behavioral therapy (CBT) intervention as part of their regular care for chronic pain showed improved function and reduced pain compared to standard treatment, according to the results of the Pain Program for Active Coping and Training (PPACT) study. Although CBT did not reduce opioid use, patients who participated in a 12-week course on pain self-management led by primary care providers showed modest but sustained benefits that persisted for 12 months after the intervention.
Study results were published this month in the Annals of Internal Medicine.
The PPACT study, an NIH Collaboratory Trial, was a pragmatic, cluster randomized trial that enrolled 850 patients receiving long-term opioid therapy for chronic pain. Patients in the intervention group participated in 12 weekly, 90-minute group sessions that taught skills of muscle relaxation, guided imagery, cognitive restructuring, and yoga-based adapted movement. Patients in the usual care group continued to receive pharmacologic and nonpharmacologic treatment.
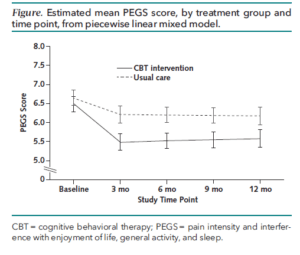
Patients were followed for 12 months with primary outcome measures of pain impact on enjoyment of life, activity levels, and sleep. Researchers also assessed secondary outcome measures of pain-related disability and opioid use.
Compared to usual care, the CBT intervention reduced self-reported pain and pain-related disability and increased satisfaction with primary healthcare providers. Opioid use and dose remained the same in both the intervention group and the usual care group.
PPACT was supported within the NIH Collaboratory by the NIH Common Fund, the National Center for Complementary and Integrative Health (NCCIH), and the National Institute of Neurological Disorders and Stroke (NINDS). Learn more about the NIH Collaboratory Trials.