Speaker
Lynn DeBar, PhD MPH
Senior Scientist
Kaiser Permanente Washington Health Research Institute
Seattle, WA
Keywords
PPACT, Chronic pain, Opioid use, Patient reported outcome data, Patient-centered research, Cognitive behavioral therapy
Key Points
- Chronic Pain is responsible for far more years lived with disability than a large number of other diseases and injuries, such as COPD, diabetes, dementia, stroke and others, combined.
- PPACT was a cluster randomized study aimed at integrating interdisciplinary pain management methods into primary care to improve care for patients with chronic pain.
- Participants had been prescribed long-term opioids for mixed chronic pain conditions.
- Study intervention lasted 12 weeks and included cognitive behavioral therapy, yoga-based exercises, physical therapy, a medication review, and support from a primary care provider.
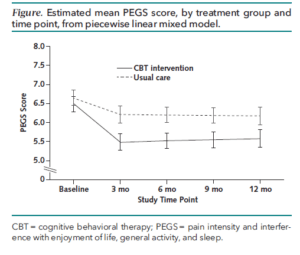
- Participants in the treatment group of the PPACT study showed a modest reduction of pain that was sustained over a 12 month period.
- The PPACT intervention was cost effective versus usual treatment methods.
Discussion Themes
An in-person enhanced enrollment session was conducted to give potential subjects a comprehensive overview of the study procedures. This enrollment session led to fewer people enrolling in the study, but of those that did, more completed the study.
The key to successful CBT is an interactive patient-centered approach.
Read more about the PPACT study.
Tags
#pctGR, @Collaboratory1