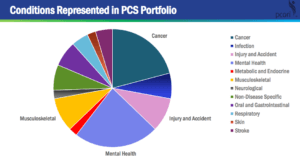
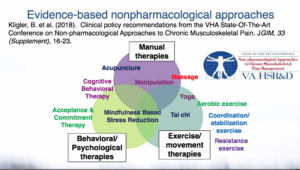
In a survey of pragmatic clinical trials across 3 NIH research networks, the complexity of delivering nonpharmacological interventions was similar between pain-related trials and non–pain-related trials. However, pain trials tended to have more intervention components, add more new tasks, and require modifications to existing workflows.
The results of the study were published online ahead of print in Contemporary Clinical Trials Communications.
The researchers surveyed study team members from trials in the NIH Pragmatic Trials Collaboratory, the IMPACT Collaboratory, and the Pain Management Collaboratory. All 3 programs support pragmatic clinical trials embedded in healthcare systems, including trials of nonpharmacological interventions for pain.
Though the trials examined in the study had similar intervention complexity, pain trials had slightly greater complexity overall, and the study teams for these trials reported needing to make more adaptations in workflows during the trial to improve the intervention’s fit or effectiveness in real-world settings.
“Change in workflow was an important consideration for intervention delivery for all trials in our study,” wrote lead author Lindsay Ballengee and her coauthors. “Future research should capture detailed, real-time information about the nature of intervention delivery complexity, adaptations, and implementation success to help improve delivery of nonpharmacologic pain interventions,” she wrote. Ballengee is a research fellow with the NIH Pragmatic Trials Collaboratory.




 The National Center for Complementary and Integrative Health (NCCIH), with support from the Helping to End Addiction Long-term® Initiative, or NIH HEAL Initiative®, published a notice of funding opportunity for pragmatic clinical trials within the infrastructure of the NIH-DOD-VA Pain Management Collaboratory (PMC).
The National Center for Complementary and Integrative Health (NCCIH), with support from the Helping to End Addiction Long-term® Initiative, or NIH HEAL Initiative®, published a notice of funding opportunity for pragmatic clinical trials within the infrastructure of the NIH-DOD-VA Pain Management Collaboratory (PMC).
 The NIH this month published notice of a funding opportunity to support the next round of pragmatic clinical trials within the NIH-DoD-VA Pain Management Collaboratory (PMC).
The NIH this month published notice of a funding opportunity to support the next round of pragmatic clinical trials within the NIH-DoD-VA Pain Management Collaboratory (PMC).