
In this Friday’s Rethinking Clinical Trials Grand Rounds, Adrian Hernandez and Rebecca Sullenger of Duke University will present “State of Clinical Trials: An Analysis of ClinicalTrials.gov.”
They will be joined by a discussion panel that will include Sara Bristol Calvert of the Clinical Trials Transformation Initiative (CTTI) and Karen Chiswell and Christopher J. Lindsell of Duke University.
The Grand Rounds session will be held on Friday, July 18, 2025, at 1:00 pm eastern.
Hernandez, who serves as co–principal investigator of the NIH Pragmatic Trials Collaboratory Coordinating Center, is a professor of medicine and vice dean in the Duke University School of Medicine and executive director of the Duke Clinical Research Institute (DCRI). Sullenger is an MD student in the Duke University School of Medicine. Calvert is director of projects at CTTI, Chiswell is a statistical scientist at the DCRI, and Lindsell is the director of data science and biostatistics at the DCRI.






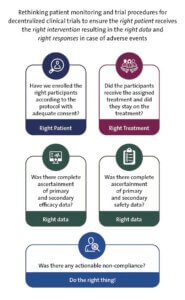 “With these risks to integrity forefront, and consistent with the principles of using decentralized elements and risk-proportionate monitoring, we propose [decentralized trials] frame their monitoring and oversight to ensure the right patient receives the right intervention, contributes the right data, and that the right response occurs for adverse events or noncompliance,” the authors wrote.
“With these risks to integrity forefront, and consistent with the principles of using decentralized elements and risk-proportionate monitoring, we propose [decentralized trials] frame their monitoring and oversight to ensure the right patient receives the right intervention, contributes the right data, and that the right response occurs for adverse events or noncompliance,” the authors wrote. In this Friday’s PCT Grand Rounds, Adrian Hernandez and Christopher Lindsell of Duke University will present
In this Friday’s PCT Grand Rounds, Adrian Hernandez and Christopher Lindsell of Duke University will present  In the latest episode of the
In the latest episode of the