Patient Engagement
Section 3
Patient Engagement Throughout a PCT
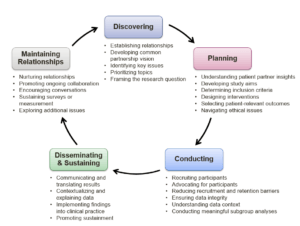
The most comprehensive patient engagement occurs throughout every step of a PCT (Figure), from developing the research concept through the dissemination of trial results and into potential sustainment. Patient engagement has become a fundamental aspect of the research. Experiences with patient engagement in research suggests that successful patient engagement can depend on patients being emotionally interested and emotionally involved in the research question. Therefore, the way the research and engagement activities are framed to potential partners is critical (Mullins et al 2012).
While targeted (ie, individual or time-limited) patient engagement activities contribute value, ongoing patient engagement allows more opportunities for patient partners to share critical insights that the research team may not have considered. Ongoing patient engagement requires more resources and time to establish relationships and trust.
A variety of frameworks are available to show different approaches to engaging patient partners. In a systematic review of frameworks for supporting patient and public involvement in research, 65 frameworks—with a variety of provenances, intended purposes, strengths, and limitations—were identified. After analysis of the many frameworks, Greenhalgh and colleagues (2019) concluded that a menu of evidence‐based resources that stakeholders can use to co‐design their own framework is the most effective.
These frameworks are also not specific to PCTs. Because of the circumstances of PCTs, it may be helpful to apply components of different frameworks and tailor the approach to the specific situation.
Figure. Patient Partner Engagement Across the PCT Cycle
Patient Engagement Methods
The method of engaging patient partners will be informed by the goals, level of engagement (as described in Section 1), resources available, and specific trial circumstances (FDA 2020). While the method of patient engagement is an important consideration, research suggests that it may not be the primary driver for the degree to which patient input influences the research (Ellis et al 2017). Just as crucial is how the researchers listen to, respond to, and incorporate the insights from patient partners with valuable lived experiences.
Some commonly used patient engagement methods include the following:
- Patient focus groups: Patient focus groups usually consist of patients “brought together to discuss a specific research issue or issues, to enhance understanding using open-ended questions” (Cambridge Biomedical Research Centre 2017). Focus groups are best facilitated by a trained moderator who follows best practices for promoting inclusivity and establishing good group dynamics (Onwuegbuzie 2009; FDA 2020).
- Community advisory boards: Community advisory boards are often composed of a diverse array of partners, including patients, practitioners, community members, policymakers, and, at times, researchers (Anderson 2021).
- Steering committee representation: A steering committee is a group intended to provide strategic direction and to have decision-making authority during the course of a PCT. It may include patients, clinicians, research coordinators, sponsors, and others.
- Individual patient interviews: Patient partners can be engaged through individual qualitative interviews with members of the research team. Ideally, the interviewer has expertise or experience in community engagement best practices and qualitative research (FDA 2020).
Dissemination
Patient partners can advise on strategies for sharing results in understandable and culturally appropriate ways. Vetting materials and means of dissemination through patient partners can help ensure effective dissemination and minimize risk of misunderstanding or misinterpretations of results.
For example, patient partners might review draft communication materials and give feedback before the materials are shared widely. They could help recommend plain language and provide guidance on making communications clear, professional, and easy to understand. When disseminating information to PCT participants, patient partners can have especially valuable insights.
Information on effectively disseminating PCT results to patients can be found in the "Dissemination to Patients" chapter. The NIH has created a Checklist for Communicating Science and Health Research to the Public, and the PCORI Dissemination and Implementation Framework provides detailed information and tools for designing and implementing a robust dissemination strategy informed by multiple partner groups.
SECTIONS
Resources
CTTI Recommendations: Patient Groups and Clinical Trials
Describes best practices for effective engagement with patient partners around clinical trials
A Guide for Focus Group, Steering Group and Advisory Panel members
Differentiates methods to engage patient partners in research
Methodological Aspects of Focus Groups in Health Research
Includes lessons learned for recruiting and communicating with focus group participants
REFERENCES
Anderson NN, Baker GR, Moody L, et al. 2021. Approaches to optimize patient and family engagement in hospital planning and improvement: Qualitative interviews. Health Expect. 24(3):967-977. doi:10.1111/hex.13239. PMID: 33761175.
Mullins CD, Abdulhalim AM, Lavallee DC. 2012. Continuous patient engagement in comparative effectiveness research. JAMA. 307(15):1587–1588. doi:10.1001/jama.2012.442. PMID: 22511684.
National Institutes for Health Research. 2017. A Guide for Focus Group, Steering Group and Advisory Panel Members. Cambridge Biomedical Research Centre. https://cambridgebrc.nihr.ac.uk/wp-content/uploads/2017/03/PPI-panel-focus-groups.pdf. Accessed January 21, 2024.
Ellis LE, Kass NE. 2017. Patient engagement in patient-centered outcomes research: challenges, facilitators and actions to strengthen the field. J Comp Eff Res.6(4):363-373. doi:10.2217/cer-2016-0075. PMID: 28621558.
Greenhalgh T, Hinton L, Finlay T, et al. 2019. Frameworks for supporting patient and public involvement in research: Systematic review and co-design pilot. Health Expect. 22(4):785-801. doi:10.1111/hex.12888. PMID: 31012259.
Onwuegbuzie AJ, Dickinson WB, Leech NL, Zoran AG. 2009. A qualitative framework for collecting and analyzing data in focus group research. International Journal of Qualitative Methods, 8(3):1-21. doi.org:10.1177/160940690900800301.



