Decentralized Pragmatic Clinical Trials
Section 4
Quality Assurance
When conducting a trial remotely and collecting data from electronic portals, smartphones, tablets, or wearables, care must be taken to ensure that it is the enrolled participant providing the information, and that data are secured in accordance with appropriate state, local, and federal regulations. The investigator may have no relationship with a patient, and the traditional chain of custody for patient data or the study interventions (be it a drug or other materials) is not replicable. Real-time participant management and communication between research teams and participants are needed to assure data quality, particularly as it relates to adherence with the study drug, outcomes ascertainment, and event monitoring.
There are critical questions for all phases of a decentralized PCT:
- Designing the trial
- Is the selected measure of health (ie, outcome) clinically meaningful and actionable?
- Enrolling participants
- How will eligibility be verified?
- How will patient identity be authenticated?
- Collecting data
- How will the identity of the person providing the data be verified?
- How will the intervention be delivered to the participant?
- How is data quality being assessed, particularly when using EHR data?
- How will lags between participants’ data entry and identification of the need for action be minimized to ensure safety and compliance, including adherence to treatment and outcome measurements?
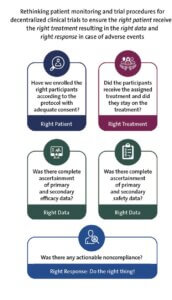
These questions indicate that investigators may need to rethink design, monitoring, and oversight of decentralized PCTs to ensure the right patient receives the right intervention, contributes the right data, and that the right response occurs for adverse events or noncompliance (Figure; Hernandez and Lindsell 2024).
Figure. The Right Patient, the Right Treatment, the Right Data, and the Right Response (Do the Right Thing!)
The Table lists some of the possible challenges that might occur during a decentralized trial, along with a list of potential solutions.
Table. Right Patient, Intervention, Data, and Procedures
| Domain | Potential Problems | Potential Solutions |
|---|---|---|
| Right patient |
|
|
| Right intervention |
|
|
| Right data |
|
|
| Right study procedures |
|
|
Patient Safety, Regulatory Issues, and Privacy Concerns
Patient safety is always a primary concern in research. While some outcomes can be assessed by remote communication with a patient, measuring functional outcomes remotely is more challenging because the clinician cannot physically interact with the patient. However, it is still possible in many settings, including via telehealth and local clinic visits. To promote patient safety, data collected via remote methods should be monitored by investigators, and not simply added to a database (Regan-Udall 2023). Additionally, self-collection of samples should be simple and non-invasive or done by specialized personnel (Vayena et al. 2023).
Another key issue for decentralized trials is a need to clarify the roles and responsibilities of healthcare providers and study investigators. For example, trials that include questionnaires that may signal patient distress or suicidal ideation must develop a plan a priori for how to respond to such signals: who is responsible and what actions should be taken to mitigate risk of harm (Ali et al. 2022). Developing a plan can be especially complicated in cases where sensitive data are collected remotely, as they may not be reviewed in real-time. Moreover, in a decentralized context, the information is unlikely to be shared with the participant’s treating clinicians. Therefore, when institutional review boards and ethics committees review decentralized trials, they will need to fully understand the timeliness of data monitoring, the specific roles and responsibilities of the researchers and clinicians, and to be certain those roles are understood by both researchers and site clinicians. If possible, it is best to ensure that the oversight bodies include individuals with expertise in decentralized trials (Vayena et al. 2023).
Regulatory ambiguity is also a concern. For example, the laws for prescribing medications across state lines are in flux. Data protections may also be ambiguous; when collecting data remotely from portals, phones, tablets, or wearables, care must be taken to ensure that data are secured in accordance with appropriate state, local, and federal regulations. The use of digital tools such as wearables and apps may increase cybersecurity risks, and appropriate safeguards should be developed to protect the privacy of participants, with special consideration given to any identifiable personal health information (Vayena et al. 2023).
Finally, with decentralized trials, some sites may be involved with certain activities of a trial without necessarily being considered “engaged in research” as a regulatory matter. Such a determination requires the entity meet the requirements of a Federalwide Assurance for the Protection of Human Subjects. The Secretary’s Advisory Committee on Human Research Protections recommends that the Office for Human Research Protections focus jurisdiction and attention on “the actual research institutions and cognizant IRBs on those entities (and their researchers) that control the primary features of study design, conduct data analysis, and communicate generalizable results to the larger scientific and public communities” (OHRP 2022).
SECTIONS
Resources
Decentralised clinical trials: ethical opportunities and challenges
This paper describes three areas requiring increased ethical vigilance: participants' safety and rights, scientific validity, and ethics oversight.
The FDA guidance provides information about decentralized trial design, remote trial visits, digital health technologies, roles and responsibilities, informed consent, and institutional review board oversight.
The EMA guidance emphasizes the importance of the patient voice and features an extensive discussion on the need for investigator oversight.
REFERENCES
Ali J, Morain SR, O’Rourke PP, Wilfond B, et al. 2022. Responding to signals of mental and behavioral health risk in pragmatic clinical trials: Ethical obligations in a healthcare ecosystem. Contemp Clin Trials. 113:106651. doi:10.1016/j.cct.2021.106651. PMID: 34998990.
Hernandez AF, Lindsell CJ. 2023. The future of clinical trials: Artificial to augmented to applied intelligence. JAMA. 330(21):2061. doi:10.1001/jama.2023.23822. PMID: 37950740.
Hernandez AF, Lindsell CJ. 2024. Ensuring virtual vigilance in decentralized clinical trials. JAMA. doi:10.1001/jama.2024.22640. PMID: 39565601.
Office of Human Research Protections (OHRP). A New Interpretation of the “Engaged in Research” Standard. 2022. Available at: https://www.hhs.gov/ohrp/sachrp-committee/recommendations/attachment-d-july-25-2022-letter/index.html. Accessed August 12, 2025.
Reagan-Udall Foundation. Good Clinical Practice: Considerations for Trials with Pragmatic or Decentralized Features. https://reaganudall.org/news-and-events/events/good-clinical-practice-considerations-trials-pragmatic-or-decentralized Accesses September 2025.
Vayena E, Blasimme A, Sugarman J. 2023. Decentralised clinical trials: Ethical opportunities and challenges. Lancet Digit Health. 5:e390–e394. doi: 10.1016/S2589-7500(23)00052-3. PMID: 37105800.
ACKNOWLEDGEMENTS
The Ethics and Regulatory Core Working Group of the NIH Collaboratory contributed to this work through discussions and edits to the text. Key contributors include Pearl O'Rourke, Joseph Ali, and Jeremy Sugarman.



