End-of-Trial Decision-Making
Section 3
Considerations for Investigators
The case studies in Section 2 show scenarios faced by investigators at the end of an ePCT’s intervention period. From the case studies, 6 themes emerged, representing factors that influence research teams’ and health system partners’ decision-making as they are approaching the end of a trial while awaiting study results (Figure 3). The themes make up a framework to help guide thoughtful decision-making about maintaining or discontinuing an ePCT intervention before clear evidence is obtained.
This framework supports researchers in identifying aspects of the waiting period that are most salient to their specific trial and relationships, thus enabling timely discussion and action planning. It can also serve as a tool to enhance transparent communication with health system partners about issues potentially influencing intervention maintenance or discontinuation, which enhances ongoing and future collaborations. Preserving trial infrastructure for the future includes not only tangible aspects of an intervention but also institutional knowledge, connections, and shared interests between researchers and health system partners. Lastly, the framework dimensions support categorizing and reflecting on experiences and lessons while approaching the end of the trial and may serve the same purpose after trial results are known.
Figure 3. End-of-Trial Decision-Making Framework
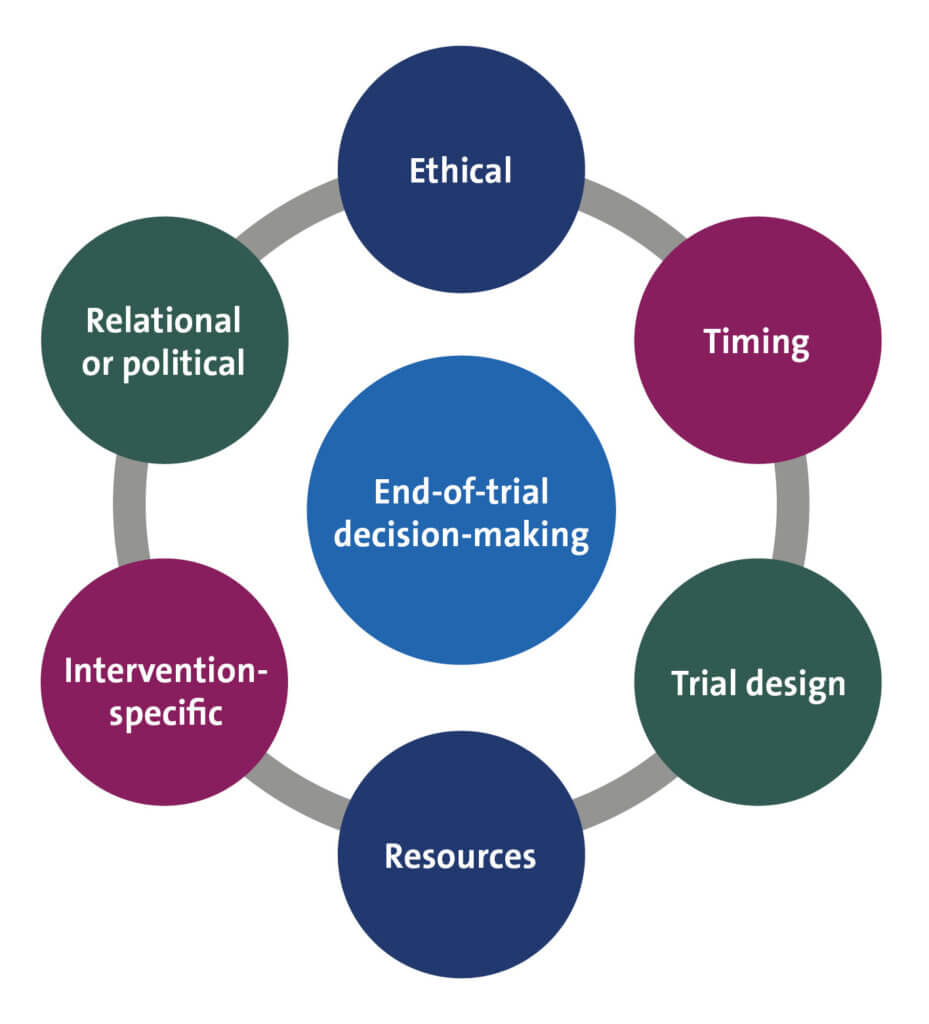
Framework Dimensions
Ethical
The ethical dimension prompts evaluating a broad set of considerations, including avoiding exploitation of trial participants, demonstrating respect for and recognition of participants’ contributions, preventing harm (such as might result from the discontinuation of a beneficial intervention), and promoting social value (a core requirement of ethical research). The particular post-trial responsibilities will vary based upon the specific features of a given PCT and are shaped by such factors as the risks to participants from discontinuation of a study intervention, the potential benefits to participants from post-trial provision of the intervention, the financial and opportunity costs associated with post-trial provision, the duration and nature of the intervention modality, and the available alternatives with similar effectiveness (Morain et al. 2024).
While historically ethics scholarship regarding post-trial responsibilities predominately focused on providing access to interventions demonstrated as beneficial (typically using the language of ‘post-trial access’), post-trial responsibilities are now understood to include a broader set of actions, such as ensuring broad access by participants and/or relevant clinical and administrative staff to study results and the resultant knowledge gained. Additionally, because health system leadership are key partners in the conduct of PCTs, they are also key partners in ensuring the appropriate fulfillment of post-trial obligations (Morain et al. 2024).
Relational or Political
Relational and political aspects of trials in the real world are central to decision-making while waiting for results. Researchers may find it difficult to keep up sustained engagement with health system partners as trial activities and perhaps funding are winding down, especially if that engagement involves somehow helping to maintain all or part of the trialed intervention. Yet, successfully cultivating such relationships may be critical to any future collaborations. Some investigators may choose to support intervention delivery as needed until the trial ends, or even after, to set the foundation for more research with the same healthcare system. One investigator noted ahead of meeting with health system partners to brainstorm new joint projects, “This is the moment when it [investment in the relationship] pays off.”
The importance of these issues extends beyond health system leadership and touches health system staff and providers involved in the trial. There may be consequences for each of these partners when maintaining or discontinuing the intervention; for example, potential employment change affecting staff who supported intervention delivery or loss of additional support for providers that was available as part of the trial intervention. Therefore, investigators should be mindful of potentially wide-ranging impacts of their activities on their health system partners.
Timing
The time that elapses between end of intervention activities and trial results can impact end-of-trial decision-making. There is a discrepancy between long research timelines and the frequently urgent problems health system leaders must address (Kush 2019; Palazzo et al. 2022). When answers are needed more quickly than trial data provide, healthcare system partners may determine to continue and eventually adopt or instead discontinue the intervention based on factors such as provider preferences and organizational priorities that are independent of trial outcomes. Finding resources to sustain an intervention until final results are available is more challenging when results are delayed by a year or more. NIH Collaboratory Trial investigators confirmed this dynamic, with one of them noting, “Timing is important. You can’t make the healthcare system wait and lose interest.”
Intervention-Specific
The nature of the intervention plays a large role in end-of-trial decisions. Experiences in the NIH Collaboratory suggest that ease of turning the intervention “on” or “off” (e.g., by activating or deactivating an EHR module) may be a determining factor. Most important for investigators is understanding whether those actions require research team involvement, and if so, to what extent. For example, research team support may be needed to discontinue the intervention in a manner that preserves essential documentation. How researchers conduct any such tasks to “exit the scene” can have important implications for relationships and possible future collaborations with the health system.
The degree of intervention adoption and integration into routine care also can affect decision-making independently of trial results. This was the case in ACP-PEACE in which one institution experienced high turnover and never fully implemented the intervention. At the opposite end of the spectrum, innovations that have become ensconced into delivery systems may be harder to abandon, especially since the degree of integration into standard work may indicate implementation success. Whether the intervention continues by inertia or by choice, investigators' involvement may be required for upkeep (e.g., to provide training to staff).
Resources
As the intervention period winds down, the research team and health system partners must determine whether resources (e.g., time, money, personnel) are available to maintain systems and intervention components enacted during the trial while their effectiveness is still unknown. Investigators may need to give special consideration to interventions that rely heavily on research team efforts for training, technical assistance, staffing, or other resource-intense aspects. If resources are at issue, it may be possible to negotiate partial support sufficient to maintain the intervention’s viability until analyses are concluded.
Other relevant factors may include the health system partner’s own resources and commitment to sustaining the intervention. For example, in ICD-Pieces, leadership turnover led to a drop in interest and dedication to research, which resulted in intervention discontinuation before results were known.
Trial Design
Various aspects of trial design have implications for end-of-trial decision making. Whether all patients or clinics within a system were recruited, or only some, influences the extent of possible repercussions of keeping or discontinuing implemented systems. With stepped-wedge designs, for example, some settings have been exposed to the intervention longer than others, which may cause tools to be integrated, and activities adopted in ways that influence choices about sustainment of the intervention.
In cluster phased rollout, changes in late-enrolling clinics’ ability to participate in the study may affect system-level decisions to maintain the intervention. Some stepped-wedge or other cluster designs may include a specific sustainment phase when support for implementation is withdrawn. In those cases, observing any decay in implementation is a design feature rather than a problem to be solved.
Decision-Making Aid
Looking across each of the framework dimensions, Table 2 offers questions investigators can consider during the end-of-trial decision-making process. These questions are also available in a downloadable worksheet.
Table 2. Dimensions Influencing End-of-Trial Decision-Making and Related Questions to Consider
| Dimension | Questions |
| Ethical |
|
| Relational or political |
|
| Timing |
|
| Intervention-specific |
|
| Resources |
|
| Trial design |
|
SECTIONS
REFERENCES
Kush RD. 2019. Learning Health Systems: Connecting Research to Practice Worldwide: Opening guest commentary. Learn Health Syst. 3(1):e10078. doi: 10.1002/lrh2.10078. PMID: 31245600.
Morain S, O'Rourke PP, Ali J, et al. 2024. Post-trial responsibilities in pragmatic clinical trials: Fulfilling the promise of research to drive real-world change. Learn Health Syst. 8(3):e10413. doi: 10.1002/lrh2.10413.
Palazzo L, Tuzzio L, Simon GE, et al. 2022. A value proposition for pragmatic clinical trials. Am J Manag Care. 28(9):e312-e314. doi: 10.37765/ajmc.2022.89224. PMID: 36121362.
ACKNOWLEDGEMENTS
The Ethics and Regulatory Core Working Group of the NIH Collaboratory contributed to this work through discussions and edits to the text. Key contributors include Stephanie Morain, Pearl O'Rourke, and Jeremy Sugarman.



