Dissemination Approaches for Different Audiences
Section 6
Dissemination to Clinicians and Health Systems
Practical Reporting Strategies for Study Teams
When planning research, by prospectively thinking about how findings will be translated to clinical practice, key considerations may be what clinicians will need to change their behavior and what resources the organization will require to permanently maintain (sustain) the intervention. — Curtis et al 2017
A key element of successful embedded pragmatic clinical trials is engagement with research partners, early and often. The research partners who will be particularly interested in the results of a pragmatic trial—and who will be crucial to the adoption of its findings—include the healthcare system leaders responsible for making decisions about which interventions to implement and sustain. Ideally, research teams will establish ongoing, bidirectional health system partnerships throughout the lifecycle of the trial and will plan their intervention with “implementation in mind.” Such planning includes considering how study results will be conveyed to the health system partners. Continuous engagement with partners can help ensure that the context for the intervention is well understood and that your partners will facilitate knowledge translation and support the intervention after the trial is completed.
The NIH Collaboratory’s Health Care Systems Interactions Core published a handout, Communicating With Health System Partners During the Lifecycle of a Trial, to provide recommendations for engaging health system leaders, clinic-level managers, and frontline staff in all phases of a study, including dissemination.
In this section, we suggest practical strategies and recommend tools for sharing study findings with healthcare system partners—beyond the usual journal publications and professional conferences. Study teams should tailor the messages and media to the needs and priorities of the health system. Make what you say clear and what you show visually appealing. Make your study results actionable, and engage with your health system partners to develop a customized dissemination plan.
Plan for Impact
From the time the proposal is written, and particularly at the start of funding, invest in relationships with healthcare system partners, including administrators and executives and frontline clinicians. Over time and with regular communication, study teams can learn the language of these leaders and staff, how the health system functions, and what level of evidence decision-makers and practitioners will use to justify their decisions. One outcome will be that health system leaders will learn the potential of your embedded intervention for improving quality and efficiency within their health system. Many researchers studying embedded interventions use the research funding or seek supplemental funding to disseminate preliminary and summative findings.
Example activities for planning for impact:
- Identify why the study findings will matter to health system leaders, whether findings are positive or negative
- Identify other partners, such as patient advocacy organizations and professional societies, that could amplify the findings if successful
- Assess the context for adopting and sustaining positive findings (see the Implementation chapter)
- Determine what study tools will be developed, including health system-, provider-, and patient-facing tools and materials
- Establish a dissemination advisory panel and consider carefully who will serve on it.
- Use a study website to share study materials, such as protocols, results summaries, and participant and research partner testimonials
It is important to disseminate findings even if the results are negative, as important learning and insight for future research can be gleaned from all results, including a hypothesized outcome that was not achieved.
The Health Foundation (United Kingdom) developed a tool kit (“Communicating Your Research”) and a template (“Developing a Communications Strategy”) to help researchers plan a communication strategy. Considerations include the context, key objectives, messaging, audience, communications channels, and additional resources.
Communicate Findings in Person
Prepare direct, in-person presentations (when possible) for your partners that synthesize your findings in an engaging format, and consider sharing results with research partners before sharing them publicly. For example, in INSPIRE, an NIH Collaboratory Trial (Gohil et al 2025a; Gohil et al 2025b), the research team presented the study’s results to the participating sites before the results were published. Research partners should also be engaged as partners in dissemination.
“Don’t write the discussion section [of your study results paper] until you’ve talked with your healthcare system partners; they might have something to teach us about what the results mean.” — Dr. Greg Simon, chair of the Health Care Systems interactions Core
Health system leaders prefer information on effectiveness over efficacy, evidence that is distilled, and results that are actionable (White et al 2017). Explain how adopting the intervention could work in their health system context. Take the time to receive feedback from stakeholders on your findings, especially what needs to be implemented or deimplemented.
Example communication products:
- Tailored briefs, executive summaries, or fact sheets
- Multimedia slide show
- Animated video
- Interactive webinar that includes polling
- Workflow demo
- Role playing
- Educational session
Provide Leave-Behind Materials and Tools
To encourage adoption of an effective intervention, study teams can leave their partner health systems with easy-to-use tools and other tangible resources. Consider devising solutions such as recommendations, clinical pathways, step-by-step instructions, and point-of-care algorithms (White et al 2017). This will help indicate that materials are already created and there is less for the health system to provide at the time of implementation and sustainability.
Example resources:
- Intervention website
- Protocol tool kit
- Animated workflow
- Infographic or poster
- Summary sheet or FAQ
- Data visualization
- Decision flowchart
- Clinical encounter aid
- Training video
- Implementation guide
- Evidence-based intervention repositories (such as the Evidence-Based Practices Resource Center, HDPulse, the Evidence-Based Cancer Control Programs [EBCCP] database, and the resources of the US Preventive Services Task Force)
SECTIONS
Resources
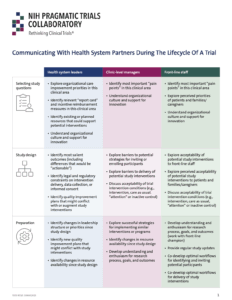
Communicating With Health System Partners During the Lifecycle of a Trial
Handout from the NIH Collaboratory’s Health Care Systems Interactions Core
Integrating Research Into Health Care Systems: Executives’ Views
Discussion paper from the National Academy of Medicine
Generating Knowledge From Best Care: Advancing the Continuously Learning Health System
Discussion paper from the National Academy of Medicine
Understanding Health Systems’ Use of and Need for Evidence to inform Decisionmaking
White paper from the Agency for Healthcare Research and Quality
A Framework for Conceptualizing Evidence Needs of Health Systems
Research white paper from the Agency for Healthcare Research and Quality
REFERENCES
Abraham E, Blanco C, Lee CC, et al. 2016. Generating Knowledge from Best Care: Advancing the Continuously Learning Health System. National Academy of Medicine, Washington, DC. https://nam.edu/perspectives/generating-knowledge-from-best-care-advancing-the-continuously-learning-health-system/. Accessed September 11, 2025.
Curtis K, Fry M, Shaban RZ, Considine J. 2017. Translating research findings to clinical nursing practice. J Clin Nurs. 26(5-6):862-872. doi: 10.1111/jocn.13586. PMID: 27649522.
Gohil SK, Septimus E, Kleinman K, et al. 2025. Improving empiric antibiotic selection for patients hospitalized with skin and soft tissue infection: The INSPIRE 3 skin and soft tissue randomized clinical trial. JAMA Intern Med. 185(6):680-691. doi: 10.1001/jamainternmed.2025.0887. PMID: 40208610.
Gohil SK, Septimus E, Kleinman K, et al. 2025. Improving empiric antibiotic selection for patients hospitalized with abdominal infection: The INSPIRE 4 cluster randomized clinical trial. JAMA Surg. 160(7):733-743. doi: 10.1001/jamasurg.2025.1108. PMID: 40208583.
Johnson K, Grossmann C, Anau J, et al. 2015. Integrating Research into Health Care Systems: Executives’ Views. Available at: https://nam.edu/perspectives-2015-integrating-research-into-health-care-systems-executives-views/. Accessed April 15, 2024.
Schoelles K, Umscheid CA, Lin JS, et al. 2017. A Framework for Conceptualizing Evidence Needs of Health Systems. Agency for Healthcare Research and Quality: Rockville, MD. https://www.ncbi.nlm.nih.gov/books/NBK493738/. Accessed September 11, 2025.
White CM, Sanders Schmidler GD, Butler M, et al. 2017. Understanding Health-Systems' Use of and Need for Evidence to Inform Decisionmaking. Agency for Healthcare Research and Quality: Rockville, MD. Report No.: 17(18)-EHC035-EF. PMID: 29611913.
ACKNOWLEDGMENTS
Karen Staman, Gina Uhlenbrauck, and Liz Wing of the NIH Pragmatic Trials Collaboratory Coordinating Center served as contributing editors for previous versions of this chapter.



