Implementation
Section 4
Implementation Frameworks
There are multiple models and conceptual frameworks for the targeted and widespread dissemination and implementation of healthcare interventions, which provide a way to descriptively organize data and concepts without attributing causal relationships (Bauer et al. 2015).
“A framework usually denotes a structure, overview, outline, system or plan consisting of various descriptive categories, e.g. concepts, constructs or variables, and the relations between them that are presumed to account for a phenomenon (Nilsen 2015).”
Theoretical approaches to implementation frameworks fall in 3 major categories, according to their overarching aims (Nilsen 2015):

A major challenge for research teams conducting pragmatic trials is selecting which model or framework will be beneficial (Zatzick et al. 2016). There are many models and frameworks for dissemination and implementation research (Tabak et al. 2012), and in this section, we will briefly describe select frameworks and introduce dissemination and implementation constructs that may be useful to investigators conducting and evaluating pragmatic trials.
Process Model Example
Action models elucidate important aspects that need to be considered in implementation practice and usually prescribe a number of stages or steps that should be followed in the process of translating research into practice. These how-to-implement models typically emphasize the importance of careful, deliberate planning, especially in the early stages of implementation endeavors (Becan et al. 2018).
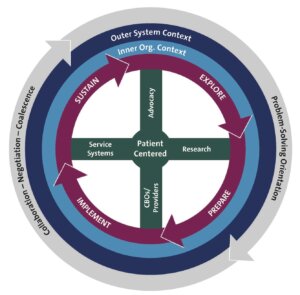
Exploration, Adoption/Preparation, Implementation, and Sustainability (EPIS) Framework
The EPIS framework articulates variables that might influence the ability to effectively implement evidence-based practices and considers both external (outer) and internal (inner or local) context of the organizations (Aarons et al. 2011).
- The exploration phase involves awareness of an issue or problem that could lead to implementing an evidence-based intervention or quality improvement approach. The outer context, at is broadest, involves state and federal funding, state legislatures, private foundations, and patient advocacy groups who support or encourage exploration of an evidence-based practice. At a local level, exploration is driven by an organization’s collective knowledge and skills, readiness for change, and receptiveness to change (Aarons et al. 2011).
- The adoption/preparation phase involves gathering and weighing research evidence. It can be driven externally by legislative changes, patient advocacy, and inter-organizational networks that may serve as partners or competitors. Adoption of an intervention is also influenced by the organization’s size, structure, and leadership (Aarons et al. 2011).
- The implementation phase is externally affected by funding, leadership, and inter-organizational networks at the level of states, counties, organizations, or groups of individuals. Additionally, the investigators who develop an intervention can help guide implementation across organizations. The internal context varies by structure (centralized vs. disperse), the priorities and goals of the organization, readiness and receptiveness for change, and the culture of the organization (Aarons et al. 2011).
- Sustainability of an intervention is driven by external leadership and policy, funding, and public academic collaborations and partnerships. At a local level, leadership and organizational culture influence sustainability, as well as the critical mass of other, possibly competing, evidence-based practices. Sustainability frequently involves fidelity monitoring and support and/or additional staffing (Aarons et al. 2011).
The Exploration, Adoption/Preparation, Implementation, and Sustainability (EPIS) Framework
Determinant Framework Example
Consolidated Framework for Implementation Research (CFIR)
CFIR was developed in 2009 (Damschroder et al. 2009) and updated in 2022. It has 5 major domains that can impact implementation to various degrees:
- Intervention characteristics: an intervention can be complex and have multiple components
- Core components are the essential elements of the intervention
- Adaptable components are elements, structures, and systems that can be changed so the intervention better fits in an organization
- Outer setting: the context of an organization and the economic, political, and social influences that are external to the actual intervention
- Inner setting: The context of an organization that directly impacts the intervention, including workflows, local culture, and structural and political influences
- Characteristics of the individuals involved
- Process of implementation
Case example: GRACE
The GRACE study was conducted to evaluate guided relaxation and acupuncture to improve pain control in individuals with chronic sickle cell disease (Doorenbos et al. 2023). One aim of the study was to “identify facilitators as well as challenges and solutions to implementing structures and processes that contribute to the seamless integration of CIH [complementary and integrative health] therapies into health care systems.” To accomplish this aim, the research team conducted qualitive implementation interviews with patients, providers and staff using interview guides that reflect the 5 domains of CFIR.
Specifically, the team interviewed
- 4 intervention participants (2 responders and 2 nonresponders) annually to elicit experiences with the intervention, perceptions of factors that hindered or facilitated participation with the intervention, aspects of the intervention they found helpful, and what might be changed.
- 10 hospital and clinic personnel (staff, providers, and interventionists) at each site to capture details about barriers and facilitators.
- At study completion, the same clinic personnel was asked to describe their experiences with integration of the interventions and to identify suggestions for the future.
Promoting Action on Research Implementation in Health Services (PARIHS)
PARIHS (Kitson et al. 1998; Ward et al. 2017) suggests that successful implementation in healthcare might be premised on 3 key determinants (characteristics of the evidence, context, and facilitation). PARIHS has subsequently undergone substantial research and development work (Ward et al. 2017) and has been widely applied (Helfrich et al. 2010).
Evaluation Framework Example
Reach, Effectiveness, Adoption, Implementation, Maintenance (RE-AIM)
Efficacy of an intervention is only a small piece of a much larger puzzle, and the “gold standard” for determining efficacy has been the explanatory clinical trial. But with PCTs, the “rigor” of trials must be balanced with “relevance” (Glasgow and Chambers 2012). Glasgow and Chambers suggest that in order to conduct research that is both rigorous and relevant, we must understand that “there is no single research design or method to identify 'truth.'” The healthcare enterprise is complex; there is much variation, and a one-size-fits-all approach of traditional research does not foster rapid dissemination and implementation of research into practice.
One factor that affects the impact of an intervention is the reach of the program (a program is the vehicle through which an intervention is delivered), which affects the percentage of the population who will receive the intervention (Abrams et al. 1996). One way to conceptualize and measure dissemination and implementation potential is the use of the RE-AIM framework, which emphasizes not only effectiveness and reach, but also adoption, implementation, and maintenance (Glasgow et al. 1999) as shown in Figure 1.
Figure. Elements of the RE-AIM Framework

Population Impact
While the core of implementation research considers implementation strategies and the associated outcomes, implementation also has a broader impact on the entire system and the health of the population. To put it another way, the public health impact of an intervention depends both on the proportion of the population who are at risk and are expected to benefit from the intervention, and on the proportion of people who are candidates for the intervention who actually receive it (Koepsell et al. 2011). With this in mind, the process of participant recruitment for a trial could provide important information about potential impact in the broader population. Expanding on this notion, effect size (a function of the trial) and reach (a function of the delivery of the intervention) have been used to project the population impact of a specific intervention (Zatzick et al. 2009).
Additionally, it is critical to consider the demand for the intervention one wishes to implement. There are two questions that bear on demand:
- Is the intervention something that is needed by a health system or provider who will be expected to implement it? Although health systems have an ethical obligation to provide treatments that work, to fully make an intervention happen, the leadership of a health system must recognize its value and be willing to devote resources—money, staff, and patient time, etc.—to it.
- Is the intervention something that patients or consumers of the intervention want and/or need? People have individual preferences and have the right to decline treatments that conventional evidence says are effective.
SECTIONS
REFERENCES
Aarons GA, Hurlburt M, Horwitz SM. 2011. Advancing a conceptual model of evidence-based practice implementation in public service sectors. Admin Policy Ment Health. 38:4–23. doi:10.1007/s10488-010-0327-7. PMID:21197565.
Abrams DB, Orleans CT, Niaura RS, Goldstein MG, Prochaska JO, Velicer W. 1996. Integrating individual and public health perspectives for treatment of tobacco dependence under managed health care: a combined stepped-care and matching model. Ann Behav Med. 18:290–304. doi:10.1007/BF02895291. PMID:18425675.
Bauer MS, Damschroder L, Hagedorn H, Smith J, Kilbourne AM. 2015. An introduction to implementation science for the non-specialist. BMC Psychol. 3:32. doi:10.1186/s40359-015-0089-9. PMID: 26376626.
Becan JE, Bartkowski JP, Knight DK, et al. 2018. A model for rigorously applying the Exploration, Preparation, Implementation, Sustainment (EPIS) framework in the design and measurement of a large scale collaborative multi-site study. Health Justice. 6:9. doi:10.1186/s40352-018-0068-3. PMID: 29654518.
Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. 2009. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 4:50. doi:10.1186/1748-5908-4-50. PMID: 19664226.
Doorenbos AZ, Schlaeger JM, deMartelly VA, et al. 2023. Hybrid effectiveness-implementation trial of guided relaxation and acupuncture for chronic sickle cell disease pain (GRACE): A protocol. Contemp Clin Trials Comm. 32:101076. doi:10.1016/j.conctc.2023.101076. PMID: 36852100.
Glasgow RE, Chambers D. 2012. Developing robust, sustainable, implementation systems using rigorous, rapid and relevant science. Clin Transl Sci. 5:48–55. doi:10.1111/j.1752-8062.2011.00383.x. PMID: 22376257.
Glasgow RE, Vogt TM, Boles SM. 1999. Evaluating the public health impact of health promotion interventions: the RE-AIM framework. Am J Public Health 89:1322–1327. PMID: 10474547.
Helfrich CD, Damschroder LJ, Hagedorn HJ, et al. 2010. A critical synthesis of literature on the promoting action on research implementation in health services (PARIHS) framework. Implement Sci. 5:82. doi:10.1186/1748-5908-5-82. PMID: 20973988.
Kitson A, Harvey G, McCormack B. 1998. Enabling the implementation of evidence based practice: a conceptual framework. Qual Health Care. 7:149–158. doi:10.1136/qshc.7.3.149. PMID: 10185141.
Koepsell TD, Zatzick DF, Rivara FP. 2011. Estimating the population impact of preventive interventions from randomized trials. Am J Prev Med 40:191–198. doi:10.1016/j.amepre.2010.10.022. PMID: 21238868.
Nilsen P. 2015. Making sense of implementation theories, models and frameworks. Implement Sci. 10:53. doi:10.1186/s13012-015-0242-0. PMID: 25895742.
Tabak RG, Khoong EC, Chambers DA, Brownson RC. 2012. Bridging research and practice. Am J Prev Med 43:337–350. doi:10.1016/j.amepre.2012.05.024. PMID: 22898128.
Ward MM, Baloh J, Zhu X, Stewart GL. 2017. Promoting Action on Research Implementation in Health Services framework applied to TeamSTEPPS implementation in small rural hospitals. Health Care Manage Rev. 42:2–13. doi:10.1097/HMR.0000000000000086. PMID: 26415078.
Zatzick DF, Koepsell T, Rivara FP. 2009. Using target population specification, effect size, and reach to estimate and compare the population impact of two PTSD preventive interventions. Psychiatry. 72:346–359. doi:10.1521/psyc.2009.72.4.346. PMID: 20070133
Zatzick DF, Russo J, Darnell D, et al. 2016. An effectiveness-implementation hybrid trial study protocol targeting posttraumatic stress disorder and comorbidity. Implement Sci 11:58. doi:10.1186/s13012-016-0424-4. PMID: 27130272



