Choosing and Specifying Endpoints and Outcomes
Section 7
Outcomes Measured via Direct Patient Report
Patients, family-members, clinicians, and researchers all want to know how treatments will improve a person’s day-to-day living and quality of life. Thus, collecting, and prioritizing the patient’s direct report on how they are feeling and functioning is often critical to evaluating the effectiveness and impact of PCTs. Patient-reported outcomes (PROs) are measured via reports taken directly from patients without interpretation by anyone else (including clinicians), and are the gold standard for assessing how people are feeling and functioning (DHHS 2025). Any PCT endpoint utilizing a PRO should be meaningful to patients and the associated measure should have appropriate validity support in the target population and setting. Additionally, other considerations are also applicable to the choice of PRO endpoints within PCTs: ideally, a PRO utilized to evaluate a PCT will have already been integrated into routine practice within clinical care settings and the data easily accessible via the electronic health record (EHR) (Zigler et al. 2024).
PRO measures reflect meaningful aspects of health and provide information about outcomes that are experienced uniquely by the patient, such as pain intensity, fatigue, and satisfaction with social roles. In an article from the NIH Pragmatic Trials Collaboratory PCO Core and EHR Core, investigators from 6 of the program’s pragmatic clinical trials shared challenges encountered for using PCOs as endpoints using the electronic health record, including
- competing healthcare system priorities
- clinician buy-in for adoption of PRO measures
- low adoption and reach of technology in low-resource settings
- lack of consensus and standardization of PRO selection and administration in the electronic health record (Zigler et al. 2024).
The authors suggest that, given the multiple barriers, study teams may need to use separate data collection systems or integrate externally collected PRO data into the electronic health record.
“When using patient-reported outcome measures for embedded pragmatic clinical trials investigators must make important decisions about whether to use data collected from the participating health system’s electronic health record, integrate externally collected patient-reported outcome data into the electronic health record, or collect these data in separate systems for their studies (Zigler et al. 2024)”
The authors developed this decision tree for using PROs in PCTs.
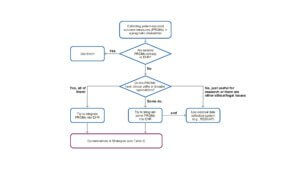
PROs still are not consistently used in clinical care, especially across different health systems and clinics, bringing unique challenges to PCTs prioritizing a patient-reported endpoint. As patient-centered outcomes become increasingly tied to quality and reimbursement, PROs are expected to be incorporated far more widely into clinical care in the coming years (Jensen et al. 2017). For now, pragmatic trials that originally plan to rely solely on data collected for billing and clinical purposes may have difficulty incorporating PROs as trial outcomes. This is not an insurmountable hurdle, however, as solutions have been developed to collect this type of information within PCTs (See the Case Study from GGC4H for an example involving RedCap.)For PRO data being extracted from the EHR, researchers planning trials should also consider the generalizability of the data across the entirety of their target population (Boyd et al. 2023).
For outcomes that represent internal sensations or experiences patients have outside the clinical visit, such as pain, symptoms, and physical functioning, the patient is the best source of information. Caregivers and other family members, especially for pediatric PCTs, can also provide important data. For other types of patient-reported health outcomes, such as co-morbidities and hospitalizations, these outcomes may also be obtained from the EHR or claims data. In such cases, the data reported by patients may supplement, contradict, or agree with EHR and claims data. For example, the EHR may not contain data on over-the-counter medications or the complete history of hospitalizations for a particular patient, and so patient report might be needed to supplement the EHR data for comprehensive outcome capture. To better understand these sources of data and how to use them in PCTs, the ADAPTABLE Supplement Report describes results of a literature review of standards for variables of interest. These deliberations resulted in a LOINC (Logical Observation Identifiers Names and Codes) patient-reported item set for ADAPTABLE.
For more on PROs, see the Living Textbook resource chapter, Patient-Reported Outcomes.
SECTIONS
Resources
Collecting patient-reported outcome measures in the electronic health record: Lessons from the NIH Pragmatic Trials Collaboratory. This paper describes best practices for collecting patient-reported outcomes.
Read more on Patient-Focused Drug Development in the Living Textbook.
Living Textbook Chapter: Patient-Reported Outcomes
This chapter describes how PROs are used in different settings and how to choose and integrate a PRO measure into an embedded pragmatic clinical trial protocol.
White Paper: Patient-Reported Outcomes
This white paper covers how to use, measure, interpret, and implement PRO measures.
REFERENCES
Boyd AD, Gonzalez-Guarda R, Lawrence K, et al. 2023. Potential bias and lack of generalizability in electronic health record data: reflections on health equity from the National Institutes of Health Pragmatic Trials Collaboratory. Journal of the American Medical Informatics Association. 30:1561–1566. doi:10.1093/jamia/ocad115.
Jensen RE, Snyder CF, Basch E, Frank L, Wu AW. 2016. All together now: findings from a PCORI workshop to align patient-reported outcomes in the electronic health record. Journal of Comparative Effectiveness Research. 5(6):561–567. PMID: 27586855 doi:10.2217/cer-2016-0026.
Patient-Focused Drug Development: Selecting, Developing, or Modifying Fit-for-Purpose Clinical Outcome Assessments. 2025.
Zigler CK, Adeyemi O, Boyd AD, et al. 2024. Collecting patient-reported outcome measures in the electronic health record: Lessons from the NIH pragmatic trials Collaboratory. Contemporary Clinical Trials. 137:107426. doi:10.1016/j.cct.2023.107426.



